New nursing home guidelines by CMS now require COVID-19 testing for nursing home staff in states with infection rates of 5% or more.
New measures, announced by the Centers for Medicare and Medicaid Services (CMS), are intended to bolster SARS-CoV-2 testing for nursing homes. These measures are also intended to provide education for staff and help decrease the spread of COVID-19 from these high-risk healthcare facilities.
With the number of COVID-19 cases around the country crossing the 5-million threshold as of Aug. 10, 2020, the new measures build on an initiative by the US Department of Health and Human Services (HHS) to provide 2,000 nursing homes around the country with rapid point-of-care (POC) SARS-CoV-2 antigen devices and 750,000 tests.
HHS designated $5 billion of Coronavirus Aid, Relief, and Economic Security (CARES) Act funds “to build nursing home skills and enhance nursing homes’ response to COVID-19, including enhanced infection control.” While the funds will go to a variety of infection control measures, providing nursing homes with the ability to perform testing on staff will be a core component of the effort.
According to HHS, “the $5 billion distribution will provide approximately $2.5 billion in upfront funding to support increased testing, staffing, and PPE needs. There will also be funding available for those establishing COVID isolation facilities. Unlike prior distributions, however, the balance of the $5 billion will be linked to nursing home performance.”
That performance will consider the prevalence of the virus in the nursing home’s local geography and will be based on the nursing home’s ability within this context to minimize COVID spread and COVID-related fatalities among its residents, according to an Aug. 7 HHS statement.
As part of the nursing home COVID-19 control emphasis, CMS announced that it will require, rather than recommend, that all nursing homes in states with a 5% positivity rate or greater test all nursing home staff each week.”
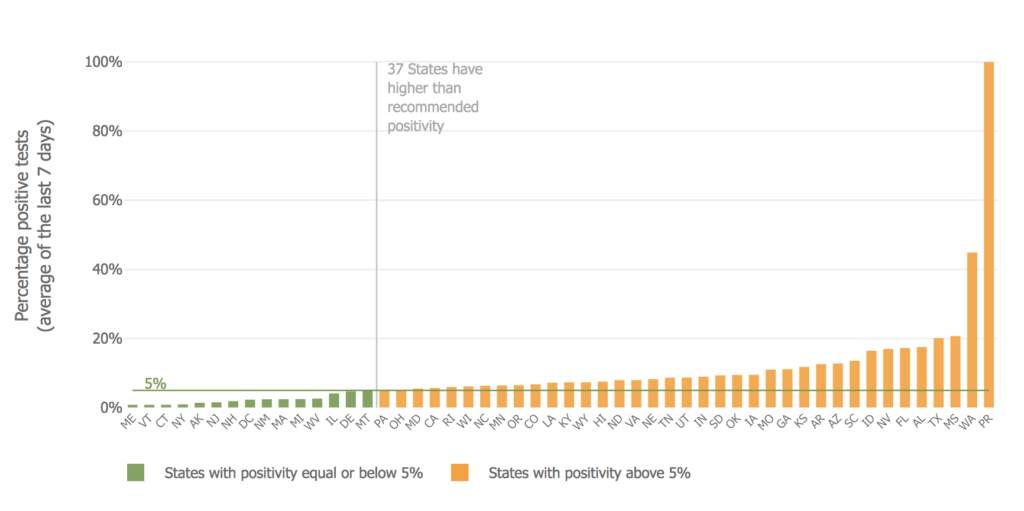
The graph above was captured Aug. 10 from the Johns Hopkins COVID Tracking Project and shows COVID-19 test positivity data by state, as gathered from publicly available data from multiple sources. In states with a higher than 5% positivity rate, CMS has recommended that nursing home staff be tested weekly for SARS-CoV-2.
As of Aug. 10, 37 states were above the recommended positivity, with the leaders then including Puerto Rico, Washington, Mississippi, Texas, Alabama, and Florida, according to the Johns Hopkins School of Medicine COVID Tracking Project.
To provide the resources necessary to perform this increased level of testing, CMS further announced, “More than 15,000 testing devices will be deployed over the next few months to help support this mandate.”
CMS Administrator Seema Verma released a statement about the nursing home COVID-19 enhanced infection control effort. “As caseloads continue to increase in areas around the country, it has never been more important that nursing homes have what they need to maintain a sturdy defense against the virus. These measures will help them do exactly that,” Verma said.
Implications for Clinical Laboratories
While there are significant new funds being allocated for SARS-CoV-2 testing in nursing homes, it seems unlikely that clinical laboratories will benefit from these new measures. In fact, the opposite may be true. If the HHS continues to fund nursing home testing on the model of this initiative, testing funds will go toward acquiring POC SARS-CoV-2 antigen test devices and test kits, allowing nursing homes to perform their own diagnostic testing and reducing PCR test volumes for local laboratories.
The push by HHS for nursing homes to have POC SARS-CoV-2 antigen testing may also indicate a continued trend toward POC antigen testing and away from the traditional PCR testing that dominated the early days of COVID-19 diagnostic testing. It remains to be seen if this trend will hold and how it could affect clinical laboratories.

—By Caleb Williams, Editor, COVID-19 STAT
Related Resources:
CMS: Trump Administration Announces New Resources to Protect Nursing Home Residents Against COVID-19
HHS Announces Allocations of CARES Act Provider Relief Fund for Nursing Homes
Wall Street Journal: Trump Administration to Mandate Covid-19 Tests for Nursing-Home Staff
The Hill: Trump administration to give additional $5B to nursing homes